Key Market Highlights
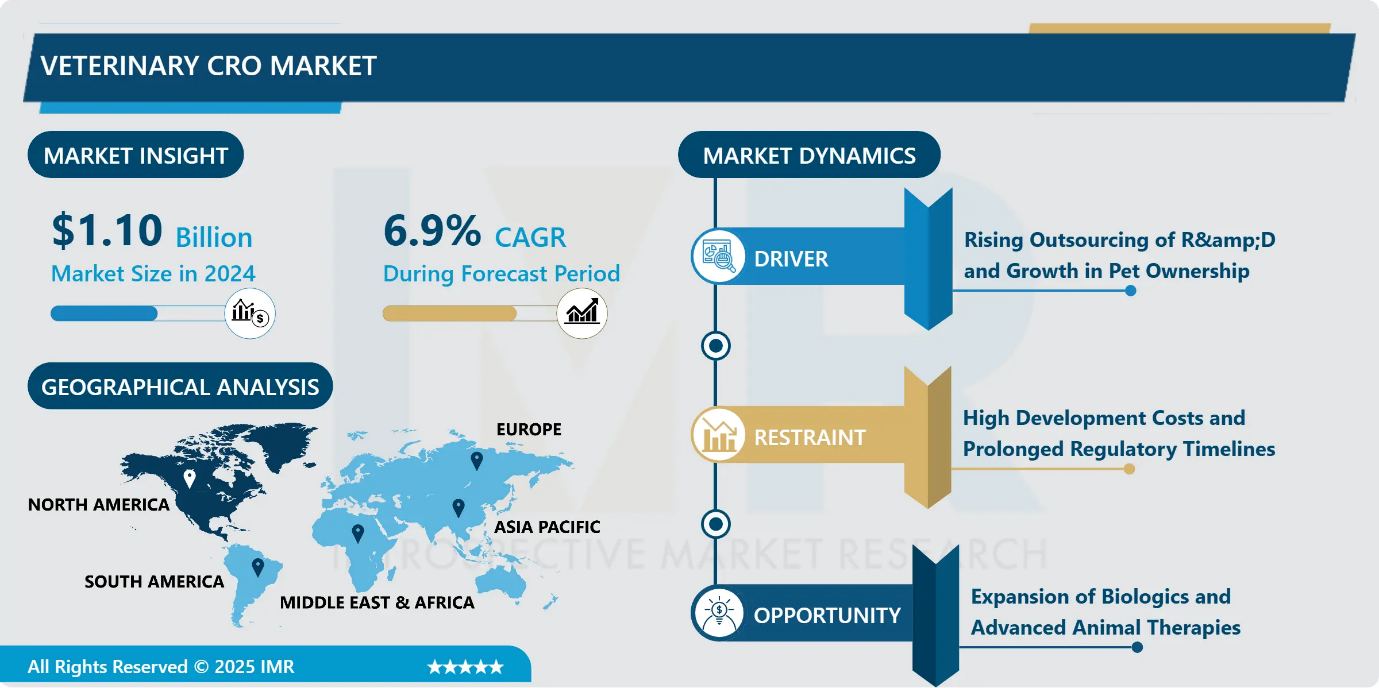
Veterinary CRO Market Size Was Valued at USD 1.10 Billion in 2024, and is Projected to Reach USD 2.29 Billion by 2035, Growing at a CAGR of 6.9% from 2025-2035.
- Market Size in 2024: USD 1.10 Billion
- Projected Market Size by 2035: USD 2.29 Billion
- CAGR (2025–2035): 6.9%
- Leading Market in 2024: North America
- Fastest-Growing Market: Asia-Pacific
- By Formulation Type: The Liquid segment is anticipated to lead the market by accounting for 27.2% of the market share throughout the forecast period.
- By End User: The Pharmaceutical and Biotechnology Companies segment is expected to capture 26.65% of the market share, thereby maintaining its dominance over the forecast period.
- By Region: North America region is projected to hold 32.9% of the market share during the forecast period.
- Active Players: Argenta (New Zealand), Aurigon Life Science (India), Avacta Animal Health (United Kingdom), Biovet (India), Charles River Laboratories (United States), Dopharma (Netherlands), and Other Active Players.
Veterinary CRO Market Synopsis:
The Veterinary CRO and CDMO Market represents a specialized outsourcing ecosystem that supports the research, development, regulatory approval, and manufacturing of animal health products. It includes contract research organizations providing preclinical studies, clinical trials, safety and efficacy testing, and regulatory consulting, alongside contract development and manufacturing organizations offering formulation development, scale-up, GMP manufacturing, and packaging services. The market is driven by rising R&D costs, growing expenditure on companion and livestock health, and increasingly stringent regulatory requirements. Sponsors are outsourcing to accelerate timelines, manage multi-country compliance, and access specialized capabilities such as vaccine challenge studies and pathogen handling. Rapid outsourcing adoption, digital trial tools, and innovation in biologics and gene-based therapies continue to expand market demand globally.
Veterinary CRO Market Dynamics and Trend Analysis:
Veterinary CRO Market Growth Driver - Rising Outsourcing of R&D and Growth in Pet Ownership
- The veterinary CRO and CDMO market is strongly driven by the increasing outsourcing of research, development, and manufacturing activities by animal health companies. Outsourcing allows sponsors to reduce capital expenditure, access specialized technical expertise, and navigate complex regulatory requirements more efficiently, thereby accelerating time to market.
- At the same time, rising global pet ownership is significantly boosting demand for advanced veterinary therapies and preventive care. As pet owners increasingly prioritize animal health and wellness, the need for innovative pharmaceuticals, biologics, and diagnostics continues to grow. This dual trend reinforces reliance on specialized CRO and CDMO partners to support scalable, cost-effective, and compliant veterinary product development.
Veterinary CRO Market Limiting Factor - High Development Costs and Prolonged Regulatory Timelines
- The veterinary CRO market faces a significant restraint due to escalating development costs and extended approval timelines for advanced animal health products, particularly biologics. Developing novel veterinary therapies requires substantial capital investment and long development cycles, driven by complex study designs, multi-site field trials, and stringent regulatory requirements. Livestock and companion-animal studies often involve high operational expenses, while extended post-approval monitoring further increases lifecycle costs. These financial and time-intensive processes limit participation by smaller innovators and concentrate project activity among well-funded sponsors. As a result, high entry barriers and prolonged timelines can slow innovation rates and constrain the overall expansion of the veterinary CRO market.
Veterinary CRO Market Expansion Opportunity - Expansion of Biologics and Advanced Animal Therapies
- The increasing development of biologics and advanced therapies in animal health presents a significant opportunity for the veterinary CRO and CDMO market. These next-generation treatments, including monoclonal antibodies, recombinant vaccines, and gene-based therapies, offer targeted and durable solutions for complex and chronic animal diseases. As the limitations of traditional small-molecule drugs become more apparent, animal health companies are increasingly relying on specialized outsourcing partners with expertise in biologics development, scale-up, and GMP manufacturing. Veterinary CROs and CDMOs play a critical role in supporting regulatory compliance, technical complexity, and innovation, creating sustained demand and long-term growth potential across companion and livestock animal segments.
Veterinary CRO Market Challenge and Risk - Limited Availability of Specialized Animal Research Infrastructure
- The veterinary CRO market faces a key challenge due to the limited availability of specialized research infrastructure required for complex animal studies. Advanced facilities capable of conducting large-animal trials under high biosafety and GLP-compliant conditions are scarce, particularly in emerging regions. This shortage often forces sponsors to relocate animals or studies across borders, increasing project timelines and operational costs.
- In addition, the high capital investment needed to establish pathogen-free herds and maintain specialized facilities discourages new infrastructure development. Limited access to advanced imaging and oncology equipment at veterinary institutions further restricts study capacity, constraining regional growth and slowing the expansion of outsourced veterinary research services.
Veterinary CRO Market Trend - Growing Emphasis on Animal Welfare and Ethical Research Practices
- The veterinary CRO market is increasingly shaped by a strong focus on animal welfare and ethical research standards. Evolving societal expectations and stricter regulatory oversight are encouraging the adoption of humane study designs, refined protocols, and ethical endpoints that minimize animal distress.
- Veterinary CROs are integrating alternative models, improved care practices, and responsible testing methodologies to align with these values. This trend is influencing study planning, execution, and client selection, as sponsors increasingly prefer partners that demonstrate ethical responsibility. CROs that prioritize animal welfare not only ensure regulatory compliance but also strengthen credibility, attract value-driven clients, and position themselves competitively in a more ethically conscious animal health research environment.
Veterinary CRO Market Segment Analysis:
Veterinary CRO Market is segmented based on into Service Type, Animal Type, Indication, Product Type, Formulation Type, End User, and Region
By Formulation Type, Liquid formulations segment is expected to dominate the market with around 27.2% share during the forecast period.
- Liquid formulations dominate the veterinary CRO market due to their ease of administration, precise dose adjustment, and broad applicability across companion and livestock animals. They are especially preferred for pets that resist solid dosage forms, improving treatment compliance and clinical outcomes. Liquid formats also simplify formulation development and stability testing, making them cost-effective for sponsors. Injectable formulations, while currently a smaller segment, are emerging rapidly driven by the growing use of biologics and the need for fast-acting therapies that bypass gastrointestinal absorption. Advances in injection technologies and rising infectious disease prevalence further support this trend. Despite injectables’ growth, liquid formulations remain dominant because of their versatility, patient compliance, and widespread clinical acceptance.
By End User, Pharmaceutical and biotechnology companies is expected to dominate with close to 26.65% market share during the forecast period.
- Pharmaceutical and biotechnology companies continued to dominate the veterinary CRO market in 2024, accounting for approximately 26.65% of total contract value. Their leadership is driven by strong financial capacity, consistent R&D pipelines, and the need to outsource complex development, regulatory, and manufacturing activities. However, academic and public research institutions are emerging as a fast-growing segment, supported by expanding One Health funding initiatives.
- These institutes are transitioning from exploratory research to conducting pivotal studies under recognized quality systems such as ISO 9001. Despite this rapid growth, industry sponsors remain dominant due to their scale, long-term outsourcing strategies, and higher volume of late-stage and commercial development projects.
Veterinary CRO Market Regional Insights:
North America region is estimated to lead the market with around 32.9% share during the forecast period.
- North America accounted for approximately 32.9% of market revenue in 2024, maintaining its dominant position due to its robust animal healthcare ecosystem. The region benefits from a dense network of GLP-certified facilities, high pet ownership levels, and strong demand for premium veterinary therapies. Favorable regulatory mechanisms, including the conditional-approval pathway offered by the U.S. Food and Drug Administration, encourage early-stage development and outsourcing.
- The presence of leading pharmaceutical and biotechnology companies, coupled with substantial R&D investments and advanced technological adoption, further strengthens market leadership. Although growth rates are moderating, entrenched infrastructure, regulatory clarity, and high spending on animal health ensure North America remains the largest and most influential regional market.
Veterinary CRO Market Active Players:
- Argenta (New Zealand)
- Aurigon Life Science (India)
- Avacta Animal Health (United Kingdom)
- Biovet (India)
- Charles River Laboratories (United States)
- Dopharma (Netherlands)
- Elanco Contract Manufacturing (United States)
- Evonik (Germany)
- Huvepharma (Bulgaria)
- LGC Group (United Kingdom)
- Lonza (Switzerland)
- MBV VET Research (Spain)
- Norbrook Laboratories (United Kingdom)
- Pharmgate (United States)
- Phibro Animal Health (United States)
- Other Active Players
Key Industry Developments in the Veterinary CRO Market:
- In January 2025, Argenta Limited announced a strategic realignment of its animal health CRO platform. The initiative is designed to strengthen end-to-end R&D and product development capabilities. It also aims to improve service integration and enhance value delivery for animal health clients.
- In April 2025, GemPharmatech expanded its U.S. footprint with the launch of a new facility in San Diego. This expansion reinforces the company’s commitment to the U.S. market. The move enhances regional preclinical research capacity and client support.
Veterinary CRO Market Technology Landscape: Preclinical, Clinical, and Regulatory Service Capabilities
- The veterinary CRO market is defined by a broad range of specialized technical capabilities supporting animal health research, development, and regulatory approval. Core services include preclinical pharmacology, toxicology, safety and efficacy studies, and clinical trials conducted across companion animals and livestock species. These studies are executed under stringent quality frameworks such as Good Laboratory Practice (GLP), Good Clinical Practice (GCP), and ISO-certified systems to meet regional and global regulatory requirements.
- Advanced study designs increasingly incorporate biomarker analysis, bioanalytical testing, digital data capture, and remote monitoring tools to improve data integrity and trial efficiency. Veterinary CROs also deploy specialized infrastructure for large-animal studies, biosafety containment, and vaccine challenge models. On the CDMO side, technical competencies extend to formulation development, process optimization, scale-up, aseptic manufacturing, and quality control testing. Ongoing adoption of biologics, gene-based therapies, and complex vaccines is further increasing technical sophistication and demand for specialized expertise across the veterinary CRO ecosystem.
|
Veterinary CRO Market |
|||
|
Base Year: |
2024 |
Forecast Period: |
2025-2035 |
|
Historical Data: |
2018 to 2024 |
Market Size in 2024: |
USD 1.10 Bn. |
|
Forecast Period 2025-32 CAGR: |
6.9 % |
Market Size in 2035: |
USD 2.29 Bn. |
|
Segments Covered: |
By Service Type |
|
|
|
By Animal Type
|
|
||
|
By Indication |
|
||
|
By Product Type |
|
||
|
By Formulation Type |
|
||
|
By End User |
|
||
|
By Region |
|
||
|
Growth Driver: |
|
||
|
Limiting Factor |
|
||
|
Expansion Opportunity |
|
||
|
Challenge and Risk |
|
||
|
Companies Covered in the Report: |
|
||
Chapter 1: Introduction
1.1 Scope and Coverage
Chapter 2:Executive Summary
Chapter 3: Market Landscape
3.1 Market Dynamics and Opportunity Analysis
3.1.1 Growth Drivers
3.1.2 Limiting Factors
3.1.3 Growth Opportunities
3.1.4 Challenges and Risks
3.2 Market Trend Analysis
3.3 Industry Ecosystem
3.4 Industry Value Chain Mapping
3.5 Strategic PESTLE Overview
3.6 Porter's Five Forces Framework
3.7 Regulatory Framework
3.8 Pricing Trend Analysis
3.9 Intellectual Property Review
3.10 Technology Evolution
3.11 Import-Export Analysis
3.12 Consumer Behavior Analysis
3.13 Investment Pocket Analysis
3.14 Go-To Market Strategy
Chapter 4: Veterinary CRO Market by Service Type (2018-2035)
4.1 Veterinary CRO Market Snapshot and Growth Engine
4.2 Market Overview
4.3 Clinical Trials and Others
4.3.1 Introduction and Market Overview
4.3.2 Historic and Forecasted Market Size in Value USD and Volume Units
4.3.3 Key Market Trends, Growth Factors, and Opportunities
4.3.4 Geographic Segmentation Analysis
Chapter 5: Veterinary CRO Market by Animal Type (2018-2035)
5.1 Veterinary CRO Market Snapshot and Growth Engine
5.2 Market Overview
5.3 Companion Animals including Dogs
5.3.1 Introduction and Market Overview
5.3.2 Historic and Forecasted Market Size in Value USD and Volume Units
5.3.3 Key Market Trends, Growth Factors, and Opportunities
5.3.4 Geographic Segmentation Analysis
5.4 Cats
5.5 and Other Companion Species
5.6 and Livestock including Cattle
5.7 Swine
5.8 Poultry
5.9 and Other Livestock
Chapter 6: Veterinary CRO Market by Indication (2018-2035)
6.1 Veterinary CRO Market Snapshot and Growth Engine
6.2 Market Overview
6.3 Oncology and Others
6.3.1 Introduction and Market Overview
6.3.2 Historic and Forecasted Market Size in Value USD and Volume Units
6.3.3 Key Market Trends, Growth Factors, and Opportunities
6.3.4 Geographic Segmentation Analysis
Chapter 7: Veterinary CRO Market by Product Type (2018-2035)
7.1 Veterinary CRO Market Snapshot and Growth Engine
7.2 Market Overview
7.3 Antimicrobials
7.3.1 Introduction and Market Overview
7.3.2 Historic and Forecasted Market Size in Value USD and Volume Units
7.3.3 Key Market Trends, Growth Factors, and Opportunities
7.3.4 Geographic Segmentation Analysis
7.4 Anti-inflammatories
7.5 Vaccines
7.6 Parasite Control Products
7.7 and Nutritional Supplements
Chapter 8: Veterinary CRO Market by Formulation Type (2018-2035)
8.1 Veterinary CRO Market Snapshot and Growth Engine
8.2 Market Overview
8.3 Liquid
8.3.1 Introduction and Market Overview
8.3.2 Historic and Forecasted Market Size in Value USD and Volume Units
8.3.3 Key Market Trends, Growth Factors, and Opportunities
8.3.4 Geographic Segmentation Analysis
8.4 Powder
8.5 Tablet
8.6 Injectable
8.7 and Topical
Chapter 9: Veterinary CRO Market by End User (2018-2035)
9.1 Veterinary CRO Market Snapshot and Growth Engine
9.2 Market Overview
9.3 Pharmaceutical and Biotechnology Companies
9.3.1 Introduction and Market Overview
9.3.2 Historic and Forecasted Market Size in Value USD and Volume Units
9.3.3 Key Market Trends, Growth Factors, and Opportunities
9.3.4 Geographic Segmentation Analysis
9.4 Veterinarians
9.5 Pet Owners
9.6 Farmers
9.7 Animal Hospitals
9.8 Research Institutions
9.9 and Others
Chapter 10: Company Profiles and Competitive Analysis
10.1 Competitive Landscape
10.1.1 Competitive Benchmarking
10.1.2 Veterinary CRO Market Share by Manufacturer/Service Provider(2024)
10.1.3 Industry BCG Matrix
10.1.4 PArtnerships, Mergers & Acquisitions
10.2 ARGENTA (NEW ZEALAND)
10.2.1 Company Overview
10.2.2 Key Executives
10.2.3 Company Snapshot
10.2.4 Role of the Company in the Market
10.2.5 Sustainability and Social Responsibility
10.2.6 Operating Business Segments
10.2.7 Product Portfolio
10.2.8 Business Performance
10.2.9 Recent News & Developments
10.2.10 SWOT Analysis
10.3 AURIGON LIFE SCIENCE (INDIA)
10.4 AVACTA ANIMAL HEALTH (UNITED KINGDOM)
10.5 BIOVET (INDIA)
10.6 CHARLES RIVER LABORATORIES (UNITED STATES)
10.7 DOPHARMA (NETHERLANDS)
10.8 ELANCO CONTRACT MANUFACTURING (UNITED STATES)
10.9 EVONIK (GERMANY)
10.10 HUVEPHARMA (BULGARIA)
10.11 LGC GROUP (UNITED KINGDOM)
10.12 LONZA (SWITZERLAND)
10.13 MBV VET RESEARCH (SPAIN)
10.14 NORBROOK LABORATORIES (UNITED KINGDOM)
10.15 PHARMGATE (UNITED STATES)
10.16 PHIBRO ANIMAL HEALTH (UNITED STATES)
10.17 AND OTHER ACTIVE PLAYERS.
Chapter 11: Global Veterinary CRO Market By Region
11.1 Overview
11.2. North America Veterinary CRO Market
11.2.1 Key Market Trends, Growth Factors and Opportunities
11.2.2 Top Key Companies
11.2.3 Historic and Forecasted Market Size by Segments
11.2.4 Historic and Forecast Market Size by Country
11.2.4.1 US
11.2.4.2 Canada
11.2.4.3 Mexico
11.3. Eastern Europe Veterinary CRO Market
11.3.1 Key Market Trends, Growth Factors and Opportunities
11.3.2 Top Key Companies
11.3.3 Historic and Forecasted Market Size by Segments
11.3.4 Historic and Forecast Market Size by Country
11.3.4.1 Russia
11.3.4.2 Bulgaria
11.3.4.3 The Czech Republic
11.3.4.4 Hungary
11.3.4.5 Poland
11.3.4.6 Romania
11.3.4.7 Rest of Eastern Europe
11.4. Western Europe Veterinary CRO Market
11.4.1 Key Market Trends, Growth Factors and Opportunities
11.4.2 Top Key Companies
11.4.3 Historic and Forecasted Market Size by Segments
11.4.4 Historic and Forecast Market Size by Country
11.4.4.1 Germany
11.4.4.2 UK
11.4.4.3 France
11.4.4.4 The Netherlands
11.4.4.5 Italy
11.4.4.6 Spain
11.4.4.7 Rest of Western Europe
11.5. Asia Pacific Veterinary CRO Market
11.5.1 Key Market Trends, Growth Factors and Opportunities
11.5.2 Top Key Companies
11.5.3 Historic and Forecasted Market Size by Segments
11.5.4 Historic and Forecast Market Size by Country
11.5.4.1 China
11.5.4.2 India
11.5.4.3 Japan
11.5.4.4 South Korea
11.5.4.5 Malaysia
11.5.4.6 Thailand
11.5.4.7 Vietnam
11.5.4.8 The Philippines
11.5.4.9 Australia
11.5.4.10 New Zealand
11.5.4.11 Rest of APAC
11.6. Middle East & Africa Veterinary CRO Market
11.6.1 Key Market Trends, Growth Factors and Opportunities
11.6.2 Top Key Companies
11.6.3 Historic and Forecasted Market Size by Segments
11.6.4 Historic and Forecast Market Size by Country
11.6.4.1 Turkiye
11.6.4.2 Bahrain
11.6.4.3 Kuwait
11.6.4.4 Saudi Arabia
11.6.4.5 Qatar
11.6.4.6 UAE
11.6.4.7 Israel
11.6.4.8 South Africa
11.7. South America Veterinary CRO Market
11.7.1 Key Market Trends, Growth Factors and Opportunities
11.7.2 Top Key Companies
11.7.3 Historic and Forecasted Market Size by Segments
11.7.4 Historic and Forecast Market Size by Country
11.7.4.1 Brazil
11.7.4.2 Argentina
11.7.4.3 Rest of SA
Chapter 12 Analyst Viewpoint and Conclusion
Chapter 13 Our Thematic Research Methodology
12.1 Research Process
12.2 Primary Research
12.3 Secondary Research
Chapter 14 Case Study
Chapter 15 Appendix
13.1 Sources
13.2 List of Tables and figures
13.3 Short Forms and Citations
13.4 Assumption and Conversion
13.5 Disclaimer
|
Veterinary CRO Market |
|||
|
Base Year: |
2024 |
Forecast Period: |
2025-2035 |
|
Historical Data: |
2018 to 2024 |
Market Size in 2024: |
USD 1.10 Bn. |
|
Forecast Period 2025-32 CAGR: |
6.9 % |
Market Size in 2035: |
USD 2.29 Bn. |
|
Segments Covered: |
By Service Type |
|
|
|
By Animal Type
|
|
||
|
By Indication |
|
||
|
By Product Type |
|
||
|
By Formulation Type |
|
||
|
By End User |
|
||
|
By Region |
|
||
|
Growth Driver: |
|
||
|
Limiting Factor |
|
||
|
Expansion Opportunity |
|
||
|
Challenge and Risk |
|
||
|
Companies Covered in the Report: |
|
||













