Global Regenerative Medicine Market Overview
Regenerative Medicine Market Size Was Valued at USD 22.32 Billion in 2023 and is Projected to Reach USD 137.53 Billion by 2032, Growing at a CAGR of 22.39% From 2024-2032
Regenerative medicine is the act of replacing or "regenerating" human cells, tissues, or organs in order to recover or restore normal function. This field has the potential to regenerate damaged tissues and organs in the body by either replacing damaged tissue or activating the body's healing processes to mend tissues or organs. Scientists may also be able to create tissues and organs in the laboratory and safely implant them when the body is unable to cure itself through regenerative medicine. Small molecule drugs, biologics, medical devices, and cell-based treatments are used in regenerative medicine. However, the word regenerative medicine is more commonly used to refer to sophisticated cell-based treatments, tissue engineering, developmental and stem cell biology, gene therapy, cellular medicines, and novel biomaterials. Regenerative medicine has shown promising results in treating several disorders from autoimmune to chronic disorders thus, fueling the growth of the market over the analysis period.
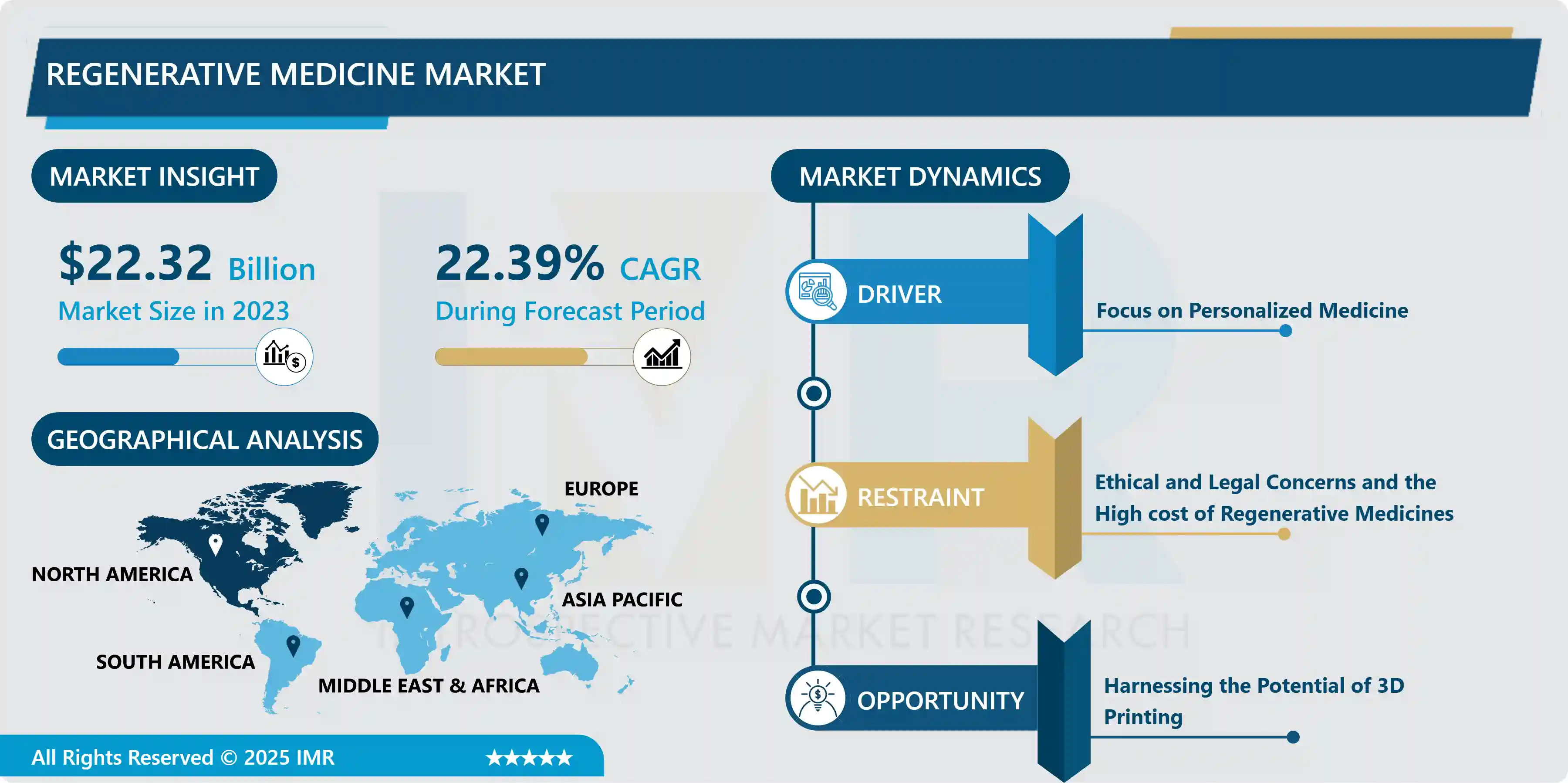
Market Dynamics And Key Factors For Regenerative Medicine Market
Drivers:
- Cord blood stem cells are being studied for a variety of applications, including Type 1 diabetes to evaluate if the cells can slow the loss of insulin production in children. In cardiovascular treatment, the cord cells are investigated to determine the migration of cells to injured cardiac tissue, improve function and blood flow at the damaged site, and thus improve the overall functioning of the heart. Cord blood stem cells are anticipated to be a valuable resource as technology moves closer to utilizing the body's cells to treat diseases. Cord blood stem cells are anticipated to be a valuable resource as technology moves closer to using the body's cells for therapy. Autologous cord blood stem cells have been an increasingly significant subject of regenerative medicine research because they can be infused back into a person without being rejected by the body's immune system.
- Regenerative medicine can lower pian by avoiding painful surgical procedures. For instance, torn rotator cuffs can be treated with cell therapy rather than surgery. Regenerative medicine therapies also reduce hospital stay time and individuals can return to their normal routine as quickly as possible. During stem cell therapy or any other regenerative medicine procedure, there is no requirement for medication or general anesthesia. Furthermore, it reduces the chances of infection that arises due to an open surgical procedure. Regenerative medicine can be utilized to treat various neurological, chronic, and autoimmune disorders thus, driving the growth of the market over the projected timeframe.
Restraints:
- The most significant impediment to the advancement of regenerative medicine is the cost. Because the cost of developing cell-based products and conducting clinical trials is so expensive, it is apparent that massive investment, both from the public and commercial sectors, is critical to the research's long-term viability. Contracts with substantial sums of money have grown in recent years as private businesses compete for this potentially valuable technology. However, the investment in regenerative medicine is risky and may incur a loss to biotechnology firms thus, restricting funding from private organizations. Although these therapies are lifesavers and more successful than traditional treatments, the response to this treatment method is less than expected thus, hampering the development of the regenerative medicine market over the forecasted timeframe.
Opportunities:
- Treatment options based on regenerative medicine have the potential to deliver long-term relief and, in some circumstances, even curative outcomes. However, if such techniques use highly tailored medicines or address narrow patient groups, reimbursement levels may need to be quite high to justify the enormous investments required to create such treatments. Traditional coding, coverage, and payment systems are often not structured to support the quick adoption or coverage of such techniques, even when they have the potential to alter medical treatment and patient outcomes. Thus, market players have to work closely with the insurance providers to evaluate the worth of these medications to meet patient demands. With the insurance companies providing coverage for this innovative treatment procedure, the demand for regenerative medicine is anticipated to increase in the forecast period thereby, creating opportunities for market players.
Market Segmentation
Segmentation Analysis of Regenerative Medicine Market:
- By product type, the cell therapy segment is expected to have the highest share of the regenerative medicine market and is projected to reach USD 25.4 billion by 2027, growing at a CAGR of 23.3% over the analysis period. Due to cardiovascular diseases, there is the formation of scar tissues which alters blood pressure or blood flow to the heart. Cell therapy can be employed in such situations and studies have shown that cell therapy can repair damaged blood vessels and heart tissues by secretion of numerous growth factors. Moreover, cell therapy can also be utilized to treat neurodegenerative diseases such as Parkinson's and Huntington's by the formation of new brain neurons, cells, and synapses after brain injuries or cognitive degeneration. Orthopedic and autoimmune disorders can also be treated with cell therapy thus bolstering the segment's growth.
- By material type, the genetically engineered tissue segment is expected to have a lion's share of the regenerative medicine market and is projected to develop at a CAGR of 21% in the forecast period. The technological advancement in molecular biology has led to the formation of new tools necessary for the design of gene-based strategies for skeletal tissue repair. Genetic engineering has emerged as a viable tool for meeting the demand for long-term and strong cellular differentiation and extracellular matrix formation. As a result, gene therapy has become a standard approach to enhancing cellular activity for skeletal tissue repair. Additionally, the increasing adoption of genetically engineered tissue products for the treatment of burns, and chronic wounds has boosted the segment's growth.
- By application, the musculoskeletal segment is predicted to dominate the regenerative medicine market during the analysis period. According to WHO, approximately 1.7 billion individuals are suffering from musculoskeletal disorders globally. Low back pain is the single largest cause of disability in 160 countries, making musculoskeletal diseases the leading contributor to disability globally. With the increasing population and aging, the prevalence of the musculoskeletal disorder is rapidly increasing. Many market players have pipeline products that can treat this disorder with stem cell therapy products. In addition, the continuous R&D to develop novel regenerative medicine for treating the musculoskeletal disorder is consolidating this segment's expansion.
Regional Analysis of Regenerative Medicine Market:
- The North American region is forecasted to have the highest share of the regenerative medicine market due to the presence of prominent market players present in this region. The regenerative medicine market was estimated at USD 3.7 billion in 2020. The Food and Drug Administration of the United States continues to enable the development and distribution of innovative medical products, such as regenerative medicine therapies, that have the potential to treat or even cure illnesses or disorders for which there are few viable treatment alternatives. The growing usage of advanced technologies to devise new treatment methods and the support by government authorities are the two main factors supporting the growth of the regenerative medicine market in this region.
- The European region is anticipated to have the second-highest share of the regenerative medicine market during the projected timeframe. The highly developed healthcare infrastructure, and the continuous research programs to evaluate the efficiency of regenerative medicine on medical disorders such as musculoskeletal, dermatology, and cardiovascular. Recent changes in the regulatory processes, coupled with an increasing political interest may improve the marketing strategies of regenerative medicine thus, strengthening the expansion of the market in the forecasted timeframe.
- The regenerative medicine market in the Asia-Pacific region is expected to develop at the highest CAGR during the analysis period. The market in this region is anticipated to reach USD 5.9 billion by the end of the projected timeframe. According to Gil Van Bokkelen, chief executive of the biotechnology company Athersys, Japan has become the center for the development of innovative therapies such as stem cells and many others. The expansion of the regenerative medicine market in this region can be attributed to the rising tissue engineering, and drug discovery programs. The growing expenditure on healthcare services and the increasing prevalence of chronic diseases have boosted the development of the market in this region.
Players Covered in Regenerative Medicine Market are:
- 3M (US)
- Orthocell Limited (Australia)
- bluebird bio (US)
- Kite Pharma (US)
- Integra LifeSciences Holdings Corporation (US)
- Spark Therapeutics (US)
- Novartis AG (Switzerland)
- Anterogen Co. Ltd. (South Korea)
- Medtronic plc (Ireland)
- Aspect Biosystems (Canada)
- Organogenesis Inc. (US)
- MiMedx Group (US)
- Misonix (US)
- Amgen Inc. (the US)
- MEDIPOST Co. Ltd. (South Korea)
- Corestem Inc. (South Korea)
Key Industry Developments in Regenerative Medicine Market
- In March 2024, NeuExcell Therapeutics, a leading biotechnology company focused on in vivo neural regenerative therapies, announced the successful dosing of the first patient with the first-in-class NeuroD1 gene therapy product NXL-004. This milestone represents significant progress in developing new treatments for malignant glioma patients.
- In December 2023, Bluebird Bio, Inc. announced its U.S. commercial infrastructure for LYFGENIA™, an FDA-approved gene therapy for patients aged 12 and older with sickle cell disease and vaso-occlusive events. The launch builds on Bluebird's success in delivering ex vivo gene therapies in the U.S. and includes outcomes-based contract offerings for payers and a personalized patient support program.
|
Global Regenerative Medicine Market |
|||
|
Base Year: |
2023 |
Forecast Period: |
2024-2032 |
|
Historical Data: |
2017 to 2023 |
Market Size in 2023: |
USD 22.32 Bn. |
|
Forecast Period 2024-32 CAGR: |
22.39% |
Market Size in 2032: |
USD 137.53 Bn. |
|
Segments Covered: |
By Product Type |
|
|
|
By Material Type |
|
||
|
By Application |
|
||
|
By Region |
|
||
|
Key Market Drivers: |
|
||
|
Key Market Restraints: |
|
||
|
Key Opportunities: |
|
||
|
Companies Covered in the report: |
|
||
Chapter 1: Introduction
1.1 Scope and Coverage
Chapter 2:Executive Summary
Chapter 3: Market Landscape
3.1 Market Dynamics
3.1.1 Drivers
3.1.2 Restraints
3.1.3 Opportunities
3.1.4 Challenges
3.2 Market Trend Analysis
3.3 PESTLE Analysis
3.4 Porter's Five Forces Analysis
3.5 Industry Value Chain Analysis
3.6 Ecosystem
3.7 Regulatory Landscape
3.8 Price Trend Analysis
3.9 Patent Analysis
3.10 Technology Evolution
3.11 Investment Pockets
3.12 Import-Export Analysis
Chapter 4: Regenerative Medicine Market by Product Type (2018-2032)
4.1 Regenerative Medicine Market Snapshot and Growth Engine
4.2 Market Overview
4.3 Cell Therapy
4.3.1 Introduction and Market Overview
4.3.2 Historic and Forecasted Market Size in Value USD and Volume Units
4.3.3 Key Market Trends, Growth Factors, and Opportunities
4.3.4 Geographic Segmentation Analysis
4.4 Gene Therapy
4.5 Tissue Engineering
4.6 Other
Chapter 5: Regenerative Medicine Market by Material Type (2018-2032)
5.1 Regenerative Medicine Market Snapshot and Growth Engine
5.2 Market Overview
5.3 Biologically Derived
5.3.1 Introduction and Market Overview
5.3.2 Historic and Forecasted Market Size in Value USD and Volume Units
5.3.3 Key Market Trends, Growth Factors, and Opportunities
5.3.4 Geographic Segmentation Analysis
5.4 Synthetic
5.5 Genetically Engineered Tissue
5.6 Other
Chapter 6: Regenerative Medicine Market by Application (2018-2032)
6.1 Regenerative Medicine Market Snapshot and Growth Engine
6.2 Market Overview
6.3 Musculoskeletal
6.3.1 Introduction and Market Overview
6.3.2 Historic and Forecasted Market Size in Value USD and Volume Units
6.3.3 Key Market Trends, Growth Factors, and Opportunities
6.3.4 Geographic Segmentation Analysis
6.4 Oncology
6.5 Cardiovascular
6.6 Dermatology
6.7 Ophthalmology
6.8 Other
Chapter 7: Company Profiles and Competitive Analysis
7.1 Competitive Landscape
7.1.1 Competitive Benchmarking
7.1.2 Regenerative Medicine Market Share by Manufacturer (2024)
7.1.3 Industry BCG Matrix
7.1.4 Heat Map Analysis
7.1.5 Mergers and Acquisitions
7.2 CARGILL INCORPORATED
7.2.1 Company Overview
7.2.2 Key Executives
7.2.3 Company Snapshot
7.2.4 Role of the Company in the Market
7.2.5 Sustainability and Social Responsibility
7.2.6 Operating Business Segments
7.2.7 Product Portfolio
7.2.8 Business Performance
7.2.9 Key Strategic Moves and Recent Developments
7.2.10 SWOT Analysis
7.3 WINDMILL ORGANICS LTDABF INGREDIENTS LTDNATURE BIO-FOODS LTD
7.4 AXIOM FOODS INCHABIB RICE PRODUCTS LTDBHARAT GLUCOSE PVT. LTDCALIFORNIA NATURAL PRODUCTS INCMATCO FOODS LIMITED
7.5 OM FOODS INCSHAFI GLUCO CHEM (PVT) LTD
7.6 WUHU DELI FOODS CO. LTDWUHU HAOYIKUAI FOOD CO. LTD
Chapter 8: Global Regenerative Medicine Market By Region
8.1 Overview
8.2. North America Regenerative Medicine Market
8.2.1 Key Market Trends, Growth Factors and Opportunities
8.2.2 Top Key Companies
8.2.3 Historic and Forecasted Market Size by Segments
8.2.4 Historic and Forecasted Market Size by Product Type
8.2.4.1 Cell Therapy
8.2.4.2 Gene Therapy
8.2.4.3 Tissue Engineering
8.2.4.4 Other
8.2.5 Historic and Forecasted Market Size by Material Type
8.2.5.1 Biologically Derived
8.2.5.2 Synthetic
8.2.5.3 Genetically Engineered Tissue
8.2.5.4 Other
8.2.6 Historic and Forecasted Market Size by Application
8.2.6.1 Musculoskeletal
8.2.6.2 Oncology
8.2.6.3 Cardiovascular
8.2.6.4 Dermatology
8.2.6.5 Ophthalmology
8.2.6.6 Other
8.2.7 Historic and Forecast Market Size by Country
8.2.7.1 US
8.2.7.2 Canada
8.2.7.3 Mexico
8.3. Eastern Europe Regenerative Medicine Market
8.3.1 Key Market Trends, Growth Factors and Opportunities
8.3.2 Top Key Companies
8.3.3 Historic and Forecasted Market Size by Segments
8.3.4 Historic and Forecasted Market Size by Product Type
8.3.4.1 Cell Therapy
8.3.4.2 Gene Therapy
8.3.4.3 Tissue Engineering
8.3.4.4 Other
8.3.5 Historic and Forecasted Market Size by Material Type
8.3.5.1 Biologically Derived
8.3.5.2 Synthetic
8.3.5.3 Genetically Engineered Tissue
8.3.5.4 Other
8.3.6 Historic and Forecasted Market Size by Application
8.3.6.1 Musculoskeletal
8.3.6.2 Oncology
8.3.6.3 Cardiovascular
8.3.6.4 Dermatology
8.3.6.5 Ophthalmology
8.3.6.6 Other
8.3.7 Historic and Forecast Market Size by Country
8.3.7.1 Russia
8.3.7.2 Bulgaria
8.3.7.3 The Czech Republic
8.3.7.4 Hungary
8.3.7.5 Poland
8.3.7.6 Romania
8.3.7.7 Rest of Eastern Europe
8.4. Western Europe Regenerative Medicine Market
8.4.1 Key Market Trends, Growth Factors and Opportunities
8.4.2 Top Key Companies
8.4.3 Historic and Forecasted Market Size by Segments
8.4.4 Historic and Forecasted Market Size by Product Type
8.4.4.1 Cell Therapy
8.4.4.2 Gene Therapy
8.4.4.3 Tissue Engineering
8.4.4.4 Other
8.4.5 Historic and Forecasted Market Size by Material Type
8.4.5.1 Biologically Derived
8.4.5.2 Synthetic
8.4.5.3 Genetically Engineered Tissue
8.4.5.4 Other
8.4.6 Historic and Forecasted Market Size by Application
8.4.6.1 Musculoskeletal
8.4.6.2 Oncology
8.4.6.3 Cardiovascular
8.4.6.4 Dermatology
8.4.6.5 Ophthalmology
8.4.6.6 Other
8.4.7 Historic and Forecast Market Size by Country
8.4.7.1 Germany
8.4.7.2 UK
8.4.7.3 France
8.4.7.4 The Netherlands
8.4.7.5 Italy
8.4.7.6 Spain
8.4.7.7 Rest of Western Europe
8.5. Asia Pacific Regenerative Medicine Market
8.5.1 Key Market Trends, Growth Factors and Opportunities
8.5.2 Top Key Companies
8.5.3 Historic and Forecasted Market Size by Segments
8.5.4 Historic and Forecasted Market Size by Product Type
8.5.4.1 Cell Therapy
8.5.4.2 Gene Therapy
8.5.4.3 Tissue Engineering
8.5.4.4 Other
8.5.5 Historic and Forecasted Market Size by Material Type
8.5.5.1 Biologically Derived
8.5.5.2 Synthetic
8.5.5.3 Genetically Engineered Tissue
8.5.5.4 Other
8.5.6 Historic and Forecasted Market Size by Application
8.5.6.1 Musculoskeletal
8.5.6.2 Oncology
8.5.6.3 Cardiovascular
8.5.6.4 Dermatology
8.5.6.5 Ophthalmology
8.5.6.6 Other
8.5.7 Historic and Forecast Market Size by Country
8.5.7.1 China
8.5.7.2 India
8.5.7.3 Japan
8.5.7.4 South Korea
8.5.7.5 Malaysia
8.5.7.6 Thailand
8.5.7.7 Vietnam
8.5.7.8 The Philippines
8.5.7.9 Australia
8.5.7.10 New Zealand
8.5.7.11 Rest of APAC
8.6. Middle East & Africa Regenerative Medicine Market
8.6.1 Key Market Trends, Growth Factors and Opportunities
8.6.2 Top Key Companies
8.6.3 Historic and Forecasted Market Size by Segments
8.6.4 Historic and Forecasted Market Size by Product Type
8.6.4.1 Cell Therapy
8.6.4.2 Gene Therapy
8.6.4.3 Tissue Engineering
8.6.4.4 Other
8.6.5 Historic and Forecasted Market Size by Material Type
8.6.5.1 Biologically Derived
8.6.5.2 Synthetic
8.6.5.3 Genetically Engineered Tissue
8.6.5.4 Other
8.6.6 Historic and Forecasted Market Size by Application
8.6.6.1 Musculoskeletal
8.6.6.2 Oncology
8.6.6.3 Cardiovascular
8.6.6.4 Dermatology
8.6.6.5 Ophthalmology
8.6.6.6 Other
8.6.7 Historic and Forecast Market Size by Country
8.6.7.1 Turkiye
8.6.7.2 Bahrain
8.6.7.3 Kuwait
8.6.7.4 Saudi Arabia
8.6.7.5 Qatar
8.6.7.6 UAE
8.6.7.7 Israel
8.6.7.8 South Africa
8.7. South America Regenerative Medicine Market
8.7.1 Key Market Trends, Growth Factors and Opportunities
8.7.2 Top Key Companies
8.7.3 Historic and Forecasted Market Size by Segments
8.7.4 Historic and Forecasted Market Size by Product Type
8.7.4.1 Cell Therapy
8.7.4.2 Gene Therapy
8.7.4.3 Tissue Engineering
8.7.4.4 Other
8.7.5 Historic and Forecasted Market Size by Material Type
8.7.5.1 Biologically Derived
8.7.5.2 Synthetic
8.7.5.3 Genetically Engineered Tissue
8.7.5.4 Other
8.7.6 Historic and Forecasted Market Size by Application
8.7.6.1 Musculoskeletal
8.7.6.2 Oncology
8.7.6.3 Cardiovascular
8.7.6.4 Dermatology
8.7.6.5 Ophthalmology
8.7.6.6 Other
8.7.7 Historic and Forecast Market Size by Country
8.7.7.1 Brazil
8.7.7.2 Argentina
8.7.7.3 Rest of SA
Chapter 9 Analyst Viewpoint and Conclusion
9.1 Recommendations and Concluding Analysis
9.2 Potential Market Strategies
Chapter 10 Research Methodology
10.1 Research Process
10.2 Primary Research
10.3 Secondary Research
|
Global Regenerative Medicine Market |
|||
|
Base Year: |
2023 |
Forecast Period: |
2024-2032 |
|
Historical Data: |
2017 to 2023 |
Market Size in 2023: |
USD 22.32 Bn. |
|
Forecast Period 2024-32 CAGR: |
22.39% |
Market Size in 2032: |
USD 137.53 Bn. |
|
Segments Covered: |
By Product Type |
|
|
|
By Material Type |
|
||
|
By Application |
|
||
|
By Region |
|
||
|
Key Market Drivers: |
|
||
|
Key Market Restraints: |
|
||
|
Key Opportunities: |
|
||
|
Companies Covered in the report: |
|
||













