Key Market Highlights
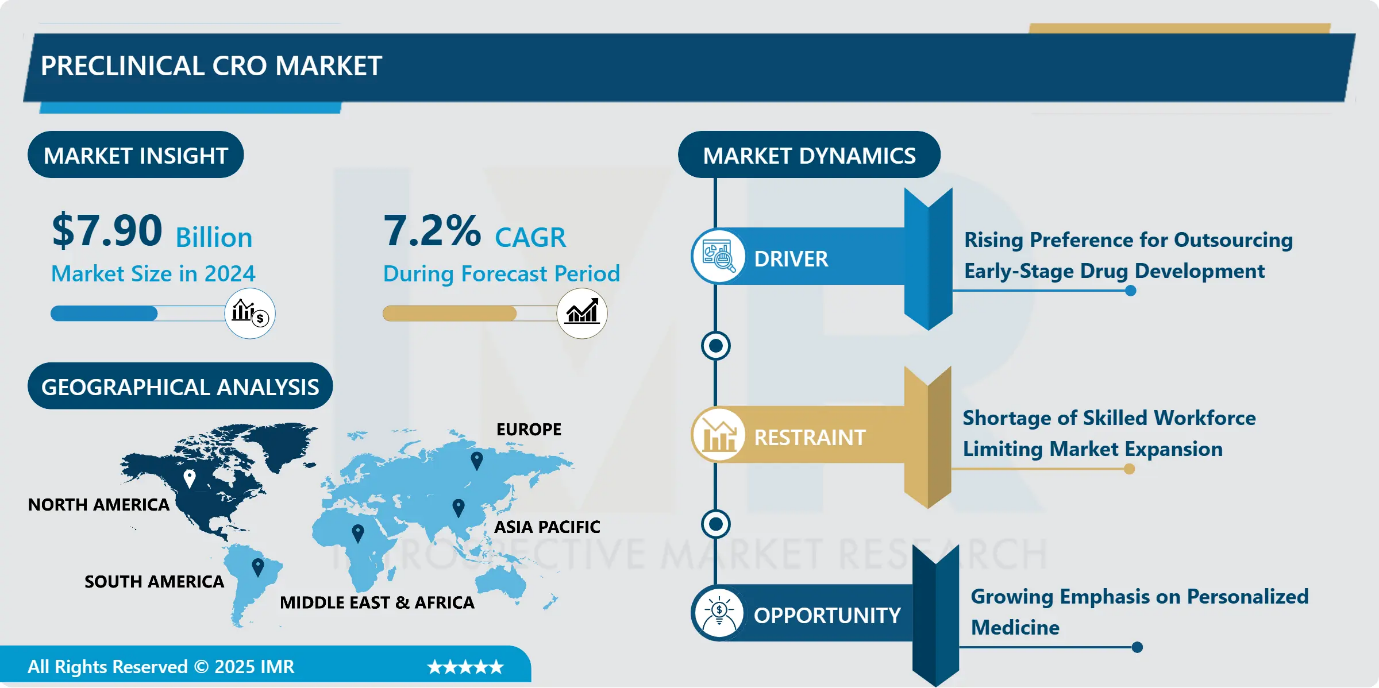
Preclinical CRO Market Size Was Valued at USD 7.90 Billion in 2024, and is Projected to Reach USD 16.97 Billion by 2035, Growing at a CAGR of 7.2% from 2025-2035.
- Market Size in 2024: USD 7.90 Billion
- Projected Market Size by 2035: USD 16.97 Billion
- CAGR (2025–2035): 7.2%
- Leading Market in 2024: North America
- Fastest-Growing Market: Asia Pacific
- By Services: The Toxicology Testing segment is anticipated to lead the market by accounting for 27.6% of the market share throughout the forecast period.
- By End User: The Biopharmaceutical Companies segment is expected to capture 37.6% of the market share, thereby maintaining its dominance over the forecast period.
- By Region: North America region is projected to hold 32.4% of the market share during the forecast period.
- Active Players: Avance Clinical (Australia), Charles River Laboratories International, Inc. (United States), Covance Inc. (United States), Crown Bioscience (United States), Envigo Corporation (United States), and Other Active Players.
Preclinical CRO Market Synopsis:
The preclinical contract research organization (CRO) market addresses a critical need within the pharmaceutical, biopharmaceutical, and medical device industries by supporting early-stage drug and device development activities. Preclinical CROs provide specialized services such as toxicology testing, pharmacokinetics, safety pharmacology, and bioanalysis, enabling sponsors to evaluate product safety and efficacy before clinical trials. The growing complexity of drug discovery, high development costs, and stringent regulatory requirements have increased reliance on outsourcing preclinical research. This market is further driven by rising R&D investments, growing innovation pipelines, and the emergence of small and mid-sized companies with limited internal infrastructure. By offering scientific expertise, advanced models, and cost efficiency, preclinical CROs accelerate development timelines and reduce financial risk for sponsors.
Preclinical CRO Market Dynamics and Trend Analysis:
Preclinical CRO Market Growth Driver - Rising Preference for Outsourcing Early-Stage Drug Development
- The growing complexity, cost, and time intensity of early-stage drug discovery are driving pharmaceutical and biotechnology companies to increasingly outsource preclinical research to specialized CROs. Outsourcing enables sponsors to optimize operational expenses, gain access to advanced scientific expertise, and accelerate development timelines. Preclinical CROs offer critical services, including pharmacology, toxicology, and efficacy studies, that support informed decision-making before clinical advancement. By leveraging external partners, drug developers can concentrate on strategic activities such as clinical development and commercialization while ensuring regulatory-compliant and high-quality preclinical outcomes. This shift toward outsourcing has emerged as a key driver supporting sustained expansion of the preclinical CRO market.
Preclinical CRO Market Limiting Factor - Shortage of Skilled Workforce Limiting Market Expansion
- The limited availability of skilled and experienced professionals represents a key restraint for the preclinical CRO market. Preclinical research involves complex R&D activities that require specialized technical expertise, regulatory knowledge, and advanced analytical capabilities. However, the industry faces a persistent shortage of qualified personnel capable of meeting these demands. This skills gap increases dependence on a limited talent pool and drives up labor costs, placing financial pressure on CROs and their clients. Elevated workforce expenses and challenges in talent acquisition can slow project execution and reduce operational efficiency, ultimately constraining the ability of preclinical CROs to scale services and sustain market growth.
Preclinical CRO Market Expansion Opportunity - Growing Emphasis on Personalized Medicine
- The increasing focus on personalized medicine is opening substantial opportunities within the preclinical CRO market. As pharmaceutical and biotechnology companies shift toward targeted and patient-specific therapies, the need for preclinical studies that reflect genetic and biological variability continues to rise. This evolution demands advanced and often complex preclinical models, including biomarker-led research and patient-derived xenograft systems. In response, preclinical CROs are strengthening their expertise in precision modeling and translational research. As personalized medicine becomes a central pillar of drug development, CROs capable of supporting customized therapeutic evaluation are likely to experience sustained demand and long-term growth.
Preclinical CRO Market Challenge and Risk - Lack of Standardization and Data Interoperability
- Limited standardization and poor data interoperability present a significant challenge in the preclinical CRO market. Inconsistent compliance with standardized data formats, such as SEND, has led to validation issues that delay regulatory reviews by authorities like the FDA and require costly resubmissions. Additionally, the widespread use of proprietary laboratory information management systems restricts seamless data integration across multiple CRO partners, increasing manual data reconciliation efforts and project costs. Fragmented metadata practices further complicate cross-study analysis, despite industry initiatives led by groups such as the Pistoia Alliance. As a result, sponsors are increasingly consolidating vendors to minimize integration complexity and operational inefficiencies.
Preclinical CRO Market Trend - Increasing Integration of Advanced Digital Technologies
- The preclinical CRO market is increasingly shaped by the integration of advanced digital technologies across research workflows. Service providers are adopting tools such as artificial intelligence and machine learning to improve data management, automate complex analyses, and accelerate study timelines. These technologies enable CROs to generate deeper insights from large preclinical datasets, enhance study design, and improve the predictability of drug safety and efficacy outcomes. As sponsors seek faster and more cost-effective development pathways, technology-driven preclinical platforms are becoming a competitive differentiator. This trend supports higher operational efficiency, better decision-making, and improved success rates in early-stage drug development.
Preclinical CRO Market Segment Analysis:
Preclinical CRO Market is segmented based on Services, Model Type, End User, and Region.
By Services, Toxicology segment is expected to dominate the market with around 27.6% share during the forecast period.
- Toxicology services remain the leading segment within the preclinical CRO market due to their mandatory role in IND-enabling programs under guidelines issued by the ICH. Repeat-dose toxicology studies across multiple species continue to be a regulatory requirement, ensuring consistent demand regardless of therapeutic area. This segment is dominant because toxicology is non-optional, highly regulated, and essential for progression into clinical development. While alternative models are gaining traction in exploratory research, they have not replaced regulatory toxicology. Safety pharmacology and DMPK services increasingly complement toxicology through integrated packages, further reinforcing toxicology’s central position in preclinical outsourcing.
By End User, Biopharmaceutical companies is expected to dominate with close to 37.6% market share during the forecast period.
- Biopharmaceutical companies represent the dominant end-user segment in the preclinical CRO market, largely due to their heavy reliance on outsourcing to enhance efficiency and manage development risk. These sponsors frequently advance multiple early-stage programs and depend on CROs for regulatory-compliant preclinical support without maintaining extensive internal infrastructure. Outsourcing enables biopharma firms to focus on innovation and clinical progression while controlling fixed costs. Although academic and research institutions are expanding their use of CRO services.
Preclinical CRO Market Regional Insights:
North America region is estimated to lead the market with around 32.4% share during the forecast period.
- North America led the global preclinical CRO market in 2024, capturing about 32.4% of total revenue. This leadership is driven by heavy pharmaceutical and biotechnology R&D investments, a dense presence of major CRO headquarters, and rigorous regulatory oversight from bodies such as the FDA and Health Canada. The region benefits from rapid adoption of advanced therapies, including biologics, cell and gene therapies, and precision medicine, all of which demand complex preclinical validation. The United States dominates within the region due to its strong innovation ecosystem, high R&D spending, and widespread outsourcing of IND-enabling studies to specialized CROs.
Preclinical CRO Market Active Players
- Avance Clinical (Australia)
- Charles River Laboratories International, Inc. (United States)
- Covance Inc. (United States)
- Crown Bioscience (United States)
- Envigo Corporation (United States)
- Eurofins Scientific (Luxembourg)
- ICON plc (Ireland)
- Intertek Group Plc (United Kingdom)
- Laboratory Corporation of America Holdings (United States)
- Medpace, Inc. (United States)
- PAREXEL International Corporation (United States)
- PPD (Thermo Fisher Scientific, Inc.) (United States)
- PRA Health Sciences, Inc. (United States)
- SGA SA (Switzerland)
- SGS SA (Switzerland)
- WuXi AppTec (China)
- Other Active Players
Key Industry Developments in the Preclinical CRO Market:
- In October 2025: Avance Clinical introduced an Early Phase Center of Excellence aimed at supporting biotech companies. The initiative is designed to streamline IND-enabling packages and accelerate early development timelines. It also focuses on delivering cost-efficient, high-quality early-phase studies.
Comprehensive Technical Framework and Regulatory-Driven Service Landscape of the Global Preclinical Contract Research Organization (CRO) Market
- The preclinical CRO market encompasses specialized service providers that support drug discovery and early development through regulated nonclinical research. These organizations conduct in vivo and in vitro studies to evaluate pharmacology, toxicology, safety pharmacology, bioanalysis, and drug metabolism and pharmacokinetics prior to first-in-human trials. Activities are performed in compliance with Good Laboratory Practice standards and aligned with regulatory expectations set by authorities such as the FDA and international guidelines issued by the ICH.
- Preclinical CROs employ advanced animal models, bioanalytical platforms, and digital data systems to generate robust, submission-ready datasets that support IND applications. Increasingly, these providers offer integrated workflows combining study design, execution, and regulatory documentation. Their technical expertise enables sponsors to manage complex modalities, ensure data integrity, and reduce development risk, making preclinical CROs a critical component of the pharmaceutical and biotechnology R&D ecosystem.
|
Preclinical CRO Market |
|||
|
Base Year: |
2024 |
Forecast Period: |
2025-2035 |
|
Historical Data: |
2018 to 2024 |
Market Size in 2024: |
USD 7.90 Bn. |
|
Forecast Period 2025-32 CAGR: |
7.2 % |
Market Size in 2035: |
USD 16.97 Bn. |
|
Segments Covered: |
By Services |
|
|
|
By Model Type
|
|
||
|
By End User |
|
||
|
By Region |
|
||
|
Growth Driver: |
|
||
|
Limiting Factor |
|
||
|
Expansion Opportunity |
|
||
|
Challenge and Risk |
|
||
|
Companies Covered in the Report: |
|
||
Chapter 1: Introduction
1.1 Scope and Coverage
Chapter 2:Executive Summary
Chapter 3: Market Landscape
3.1 Market Dynamics and Opportunity Analysis
3.1.1 Growth Drivers
3.1.2 Limiting Factors
3.1.3 Growth Opportunities
3.1.4 Challenges and Risks
3.2 Market Trend Analysis
3.3 Industry Ecosystem
3.4 Industry Value Chain Mapping
3.5 Strategic PESTLE Overview
3.6 Porter's Five Forces Framework
3.7 Regulatory Framework
3.8 Pricing Trend Analysis
3.9 Intellectual Property Review
3.10 Technology Evolution
3.11 Import-Export Analysis
3.12 Consumer Behavior Analysis
3.13 Investment Pocket Analysis
3.14 Go-To Market Strategy
Chapter 4: Preclinical CRO Market by Services (2018-2035)
4.1 Preclinical CRO Market Snapshot and Growth Engine
4.2 Market Overview
4.3 Toxicology Testing
4.3.1 Introduction and Market Overview
4.3.2 Historic and Forecasted Market Size in Value USD and Volume Units
4.3.3 Key Market Trends, Growth Factors, and Opportunities
4.3.4 Geographic Segmentation Analysis
4.4 Safety Pharmacology
4.5 Drug Metabolism
4.6 Pharmacokinetics
4.7 IND-Enabling Programs
4.8 Bioanalysis & DMPK Studies
4.9 and Other Services
Chapter 5: Preclinical CRO Market by Model Type (2018-2035)
5.1 Preclinical CRO Market Snapshot and Growth Engine
5.2 Market Overview
5.3 Patient-Derived Organoid (PDO) Models
5.3.1 Introduction and Market Overview
5.3.2 Historic and Forecasted Market Size in Value USD and Volume Units
5.3.3 Key Market Trends, Growth Factors, and Opportunities
5.3.4 Geographic Segmentation Analysis
5.4 Patient-Derived Xenograft (PDX) Models
5.5 and In-Silico / AI-Driven Models
Chapter 6: Preclinical CRO Market by End User (2018-2035)
6.1 Preclinical CRO Market Snapshot and Growth Engine
6.2 Market Overview
6.3 Biopharmaceutical Companies
6.3.1 Introduction and Market Overview
6.3.2 Historic and Forecasted Market Size in Value USD and Volume Units
6.3.3 Key Market Trends, Growth Factors, and Opportunities
6.3.4 Geographic Segmentation Analysis
6.4 Medical Device Manufacturing Companies
6.5 Academic and Research Organizations
6.6 and Other End Users
Chapter 7: Company Profiles and Competitive Analysis
7.1 Competitive Landscape
7.1.1 Competitive Benchmarking
7.1.2 Preclinical CRO Market Share by Manufacturer/Service Provider(2024)
7.1.3 Industry BCG Matrix
7.1.4 PArtnerships, Mergers & Acquisitions
7.2
7.2.1 Company Overview
7.2.2 Key Executives
7.2.3 Company Snapshot
7.2.4 Role of the Company in the Market
7.2.5 Sustainability and Social Responsibility
7.2.6 Operating Business Segments
7.2.7 Product Portfolio
7.2.8 Business Performance
7.2.9 Recent News & Developments
7.2.10 SWOT Analysis
Chapter 8: Global Preclinical CRO Market By Region
8.1 Overview
8.2. North America Preclinical CRO Market
8.2.1 Key Market Trends, Growth Factors and Opportunities
8.2.2 Top Key Companies
8.2.3 Historic and Forecasted Market Size by Segments
8.2.4 Historic and Forecast Market Size by Country
8.2.4.1 US
8.2.4.2 Canada
8.2.4.3 Mexico
8.3. Eastern Europe Preclinical CRO Market
8.3.1 Key Market Trends, Growth Factors and Opportunities
8.3.2 Top Key Companies
8.3.3 Historic and Forecasted Market Size by Segments
8.3.4 Historic and Forecast Market Size by Country
8.3.4.1 Russia
8.3.4.2 Bulgaria
8.3.4.3 The Czech Republic
8.3.4.4 Hungary
8.3.4.5 Poland
8.3.4.6 Romania
8.3.4.7 Rest of Eastern Europe
8.4. Western Europe Preclinical CRO Market
8.4.1 Key Market Trends, Growth Factors and Opportunities
8.4.2 Top Key Companies
8.4.3 Historic and Forecasted Market Size by Segments
8.4.4 Historic and Forecast Market Size by Country
8.4.4.1 Germany
8.4.4.2 UK
8.4.4.3 France
8.4.4.4 The Netherlands
8.4.4.5 Italy
8.4.4.6 Spain
8.4.4.7 Rest of Western Europe
8.5. Asia Pacific Preclinical CRO Market
8.5.1 Key Market Trends, Growth Factors and Opportunities
8.5.2 Top Key Companies
8.5.3 Historic and Forecasted Market Size by Segments
8.5.4 Historic and Forecast Market Size by Country
8.5.4.1 China
8.5.4.2 India
8.5.4.3 Japan
8.5.4.4 South Korea
8.5.4.5 Malaysia
8.5.4.6 Thailand
8.5.4.7 Vietnam
8.5.4.8 The Philippines
8.5.4.9 Australia
8.5.4.10 New Zealand
8.5.4.11 Rest of APAC
8.6. Middle East & Africa Preclinical CRO Market
8.6.1 Key Market Trends, Growth Factors and Opportunities
8.6.2 Top Key Companies
8.6.3 Historic and Forecasted Market Size by Segments
8.6.4 Historic and Forecast Market Size by Country
8.6.4.1 Turkiye
8.6.4.2 Bahrain
8.6.4.3 Kuwait
8.6.4.4 Saudi Arabia
8.6.4.5 Qatar
8.6.4.6 UAE
8.6.4.7 Israel
8.6.4.8 South Africa
8.7. South America Preclinical CRO Market
8.7.1 Key Market Trends, Growth Factors and Opportunities
8.7.2 Top Key Companies
8.7.3 Historic and Forecasted Market Size by Segments
8.7.4 Historic and Forecast Market Size by Country
8.7.4.1 Brazil
8.7.4.2 Argentina
8.7.4.3 Rest of SA
Chapter 9 Analyst Viewpoint and Conclusion
Chapter 10 Our Thematic Research Methodology
9.1 Research Process
9.2 Primary Research
9.3 Secondary Research
Chapter 11 Case Study
Chapter 12 Appendix
10.1 Sources
10.2 List of Tables and figures
10.3 Short Forms and Citations
10.4 Assumption and Conversion
10.5 Disclaimer
|
Preclinical CRO Market |
|||
|
Base Year: |
2024 |
Forecast Period: |
2025-2035 |
|
Historical Data: |
2018 to 2024 |
Market Size in 2024: |
USD 7.90 Bn. |
|
Forecast Period 2025-32 CAGR: |
7.2 % |
Market Size in 2035: |
USD 16.97 Bn. |
|
Segments Covered: |
By Services |
|
|
|
By Model Type
|
|
||
|
By End User |
|
||
|
By Region |
|
||
|
Growth Driver: |
|
||
|
Limiting Factor |
|
||
|
Expansion Opportunity |
|
||
|
Challenge and Risk |
|
||
|
Companies Covered in the Report: |
|
||













